
CE/ISO/GMP Soft Bag IV
Description
Basic Info.
| Certification | CE, GMP, ISO |
| Voltage | 380V |
| Computerized | Computerized |
| Customized | Customized |
| Condition | New |
| Warranty | >2 Year |
| After-sales Service | Video Online, Field Installation |
| Application | Chemical, Medical, Food |
| Transport Package | Export Wooden Case |
| Specification | Standard/customized |
| Trademark | Marya |
| Origin | China |
| HS Code | 8414301501 |
| Production Capacity | 5000pieces/Year |
Product Description

CE/ISO/GMP Soft Bag IV-Solution Production Line Pharmaceutical Filling Machine Production Line
The Marya Pharmaceutical Soft Bag Forming Printing Filling Sealing IV Production Machine in Shanghai has the characteristics of innocuity, strong compatibility with a drug solution, good isolating air, and water, long keeping period, resistant high-temperature,anti-low temperature, and convenient transportation. In the process of infusion, there is no need to add an inlet tube due to the automatic contract of the soft bag, eliminating the second solution during infusion, recycling empty bag does not pollute the environment.
1.Film feeding
Adopt a constant tension system, making the tension force of the film stable.
2.Film stretching
Adopt continuous film stretching technology, which saves 1 second of time comparing with the original one and promotes the production line speed.
3.Lettering
Adopt thermal transfer technology, and print the content on the printing plate to the film surface.
4.Port feeding
It is equipped with a bag storage hopper, ensuring that there is always a small amount of bag storage in the feeding shock plate and can conduct automatic monitoring and feeding, to prevent particles occurring when the material has mutual friction in the feeding.
5.Preheating
The temperature ensures the preheating molds are at the same temperature.
6.Forming
The heating pipe is directly installed in the mold, the temperature probe is installed on the forming mold surface, the accuracy of forming temperature control is high, and the phenomenon of inconsistency between the forming temperature and the displayed temperature shall not appear, so as to ensure the high qualified rate.
7.Port welding, film cooling, and chamfering
Each welding mold was driven by a single-cylinder, the adaptability of the port is strong, and it shall not influence the welding quality between the port and film due to change of the overall dimension of the port;
Features
Model Item | MSRD2500 | MSRD5000 | MSRD7500 |
Max Production Capacity(b/h) | 2500 | 5000 | 7500 |
Bag-making number each time | 2 | 4 | 6 |
Filling and sealing number each time | 2 | 4 | 6 |
Electric capacity(kw/v/Hz) | 25/380/50 | 32/380/50 | 43/380/50 |
(0.6Mpa)dry,oil-free compressed air consumption(L/min) | 2000 | 4000 | 5000 |
(0.4Mpa)clean air consumotion(L/min) | 800 | 1500 | 2000 |
Cooling water consumption | 15-20ºC 300L/h | ||
Filling drug solution | T≤50ºC 0.1-0.2Mpa | ||
Bag making film | Non-PVC double layer flat film Film thickness:190±20um | ||
Film canister size | Inner diameter:Ø150; outer diamerer of film canister≤Ø600 | ||
Bag making shape | Single tube, heat welding sealing | ||
Printing Method | Heat transferring printing or intelligent foil printing, bar-code printing | ||
The inner diameter for ribbon canister | Ø76 | ||
Noise | <75dB | ||
Main mater | 304 stainless steel.The contact part with drug solution is 316L,which conforms to required material of GMP | ||
Specification | 50ml,100ml,250ml,500ml,1000ml | 100ml,250ml,500ml | |
Weigt | 5000kg | 7000kg | 8000kg |
Overall dimension(L×W×H) | 7650×2335×2500 | 9600×2250×2400 | 8400×2880×2140 |
About Us
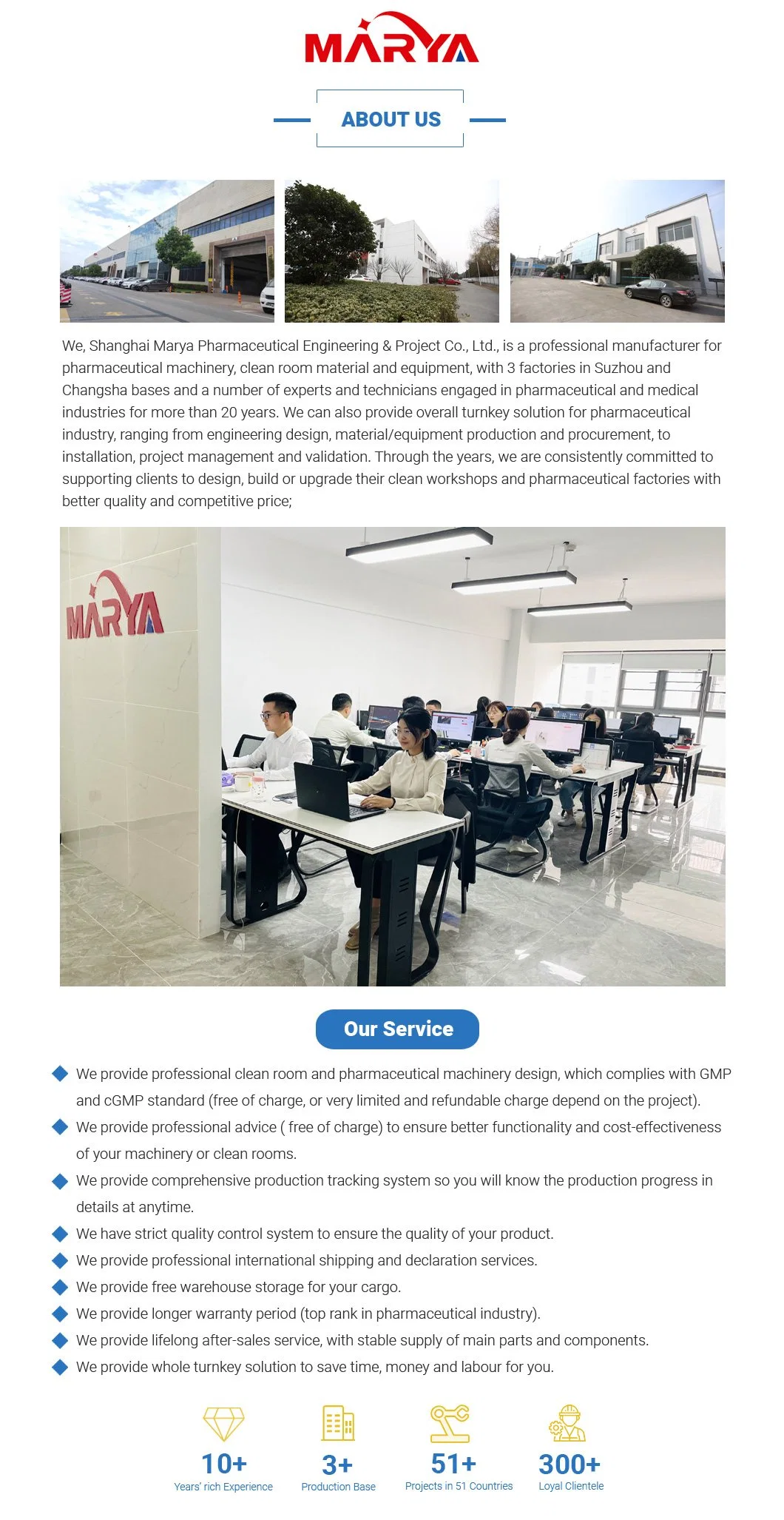
Workshop

Certificate

Project case

Our Customer
Packaging & Shipping
CE/ISO/GMP Soft Bag IV-Solution Production Line Pharmaceutical Filling Machine Production Line
Our Contact






